Infectious Disease Division faculty perform clinical as well as basic science research. Close scientific collaborative relationships exist with the Department of Immunobiology, Division of Pulmonary, Allergy, Crticial Care and Sleep Medicine, and Mel and Enid Zuckerman College of Public Health.
Important resources available to researchers at the University of Arizona College of Medicine – Tucson include a diagnostic laboratory; biostatistical, bioinformatics and computing core services; flow cytometry, microscopy, genomics and proteomics cores; a clinical translational science center that supports clinical trials; and the Sarver Heart Center; the Asthma and Airway Disease Research Center; the Arizona Center on Aging; and the Arizona Cancer Center — a National Cancer Institute (NCI)-Designated Comprehensive Cancer Center — as well as other centers and institutes both related to the Department of Medicine and UArizona Health Sciences.
UArizona Infectious Diseases Research Investigator Profiles
HIV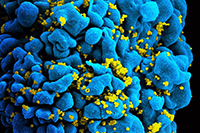
Scanning electromicrograph (right) of an HIV-infected T cell.
A variety of HIV-related research is performed by division faculty ranging from clinical studies of optimal implementation of Pre-exposure Prophylaxis (PrEP) and other interventions to reduce coronary artery disease, to basic immunology research aimed at finding a cure for HIV infection. Many of these studies draw upon the patient population that is served by the Petersen HIV Clinics.
Valley Fever (Coccidioidomycosis)
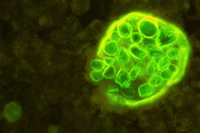 Spherule of Coccidioides immitis (left) with endospores.
Spherule of Coccidioides immitis (left) with endospores.
One of the missions of the Valley Fever Center for Excellence is to promote research that will improve the care of people with Valley fever (also known as coccidioidomycosis, or cocci) and eventually prevent the disease. Ongoing studies include a clinical trial of empiric treatment for Valley fever along with routine treatment of community acquired pneumonia, as well as more basic studies to investigate the genetics associated with disseminated disease and to develop a vaccine against coccidioidomycosis—both in animals and humans.
Kissing Bugs
 Triatominae (right), a subfamily of Reduviidae, are known also as conenose, kissing, assassin or vampire bugs.
Triatominae (right), a subfamily of Reduviidae, are known also as conenose, kissing, assassin or vampire bugs.
Kissing bug bites can trigger serious allergic reactions in humans as well as transmit disease to people and their pets. Bugs infected with the parasite, Trypanosoma cruzi, can cause Chagas disease, which has two phases, acute and chronic. Acute symptoms include fever, chills, fatigue, body aches, nausea, diarrhea or vomiting within several weeks of a bite. One-third of people with Chagas may suffer chronic issues that may lead to an enlarged heart or intestinal cavity, which is why it’s important to catch symptoms early. Eleven species of kissing bugs exist across 28 U.S. states, including Arizona. Among ongoing projects are an effort to determine and evaluate the emergence of a kissing bug species, Triatoma recurva, in Bisbee, Arizona, and monitoring kissing bug home intrusion in Tucson.
Transplant Infections
Our Transplant Infectious Diseases program, led by Tirdad Zangeneh, MD, is dedicated to improving the care of transplant patients through research. Ongoing studies are aimed at understanding the influence of different therapies on clinical outcomes of invasive fungal infections, the prevalence and clinical outcomes of de novo coccidiodomycosis infections in solid organ transplant patients, a meta-analysis of treatment outcomes in drug-resistant CMV (Cytomegalovirus) infections, and predisposing factors and clinical outcomes of nocardiosis in solid organ transplant patients.
Current Research Studies
For UArizona research studies, including those currently accepting clinical subjects, see the following topics related to our Division:
 “Pathogenesis and Genetics of Disseminated or Refractory Coccidioidomycosis”
“Pathogenesis and Genetics of Disseminated or Refractory Coccidioidomycosis”
Investigator: John Galgiani, MD
Coccidioidomycosis is caused by a fungus that grows in the U.S. Southwest, parts of Mexico and South America. This disease is caused by breathing dust containing the fungus. Infection can lead to serious lung and breathing problems. When it spreads outside the lungs, this is called disseminated coccidioidomycosis (DCM). If the fungus stays in the lungs for more than 6 months, it is called refractory coccidioidomycosis (RCM). People with DCM or RCM may have difficulty fighting off infection because of immune system problems. Researchers want to study the immune systems of people with DCM or RCM, to learn more about the disease and the best ways to treat it.
“IMMUNO-GENETIC BASIS FOR HUMAN DISSEMINATED COCCIDIOIDOMYCOSIS”
Investigators: John Galgiani, MD, and Yves Lussier, MD (right)
This project permits investigators at the National Institutes of Health and the UArizona to work together, utilizing unique resources of each institution. Findings from these studies may provide new tests to determine which persons are at risk of serious disease if they contract Valley Fever. They may also help in the development of preventative vaccines.
 “EARLY FLUCONAZOLE TREATMENT FOR COCCIDIOiDOMYCOSIS PNEUMONIA”
“EARLY FLUCONAZOLE TREATMENT FOR COCCIDIOiDOMYCOSIS PNEUMONIA”
Investigators: Fariba M. Donovan, MD Phd and Aneesh Narang, MD
This is an observational study in up to 1,000 individuals age 18 or older, with community-acquired pneumonia. It's designed to provide data on effectiveness of early management strategies for coccidioidomycosis pneumonia (Valley fever pneumonia or acute onset Valley fever).
 “AN OBSERVATIONAL DISEASE REGISTRY OF PATIENTS TREATED WITH SYSTEMIC MOLD-ACTIVE TRIAZOLES”
“AN OBSERVATIONAL DISEASE REGISTRY OF PATIENTS TREATED WITH SYSTEMIC MOLD-ACTIVE TRIAZOLES”
Investigator: Tirdad Zangeneh, DO (on right at right)
This study seeks to describe representative real-world patterns of care for the management of invasive fungal infections (IFIs), including invasive mold infection (IMI). Specifically, the study goals are to examine real world patient characteristics and treatment patterns, associated healthcare resource utilization, and outcomes associated with use of mold-active triazoles (MATs) to treat IFIs.
“Non Interventional Registry Study of Systemic Antifungal Therapy in Adult Subjects with Invasive Mucormycosis or Invasive Aspergillosis Caused by a Non fumigatus Species”
Investigator: Tirdad Zangeneh, DO
The objective of this study is to obtain and report outcome data for adult patients who received systemic antifungal therapy (AFT) for the treatment of invasive mucormycosis (IM) or invasive aspergillosis (IA) caused by a non-fumigatus species.
 “Pre-Exposure Prophylaxis (PrEP) Knowledge, Attitudes, and Barriers Among Individuals with Limited Access to Medical Care and from Diverse Ethnic Backgrounds”
“Pre-Exposure Prophylaxis (PrEP) Knowledge, Attitudes, and Barriers Among Individuals with Limited Access to Medical Care and from Diverse Ethnic Backgrounds”
Investigator: Lori Fantry, MD, MPH
“Pathogenesis of Rebound SIV/HIV Viremia After Antiretroviral Therapy"
Investigator: Elizabeth Connick, MD
“The Cost of Delays in Diagnosing Early Coccidioidal Infection at BUMC Tucson”
Investigator: Fariba Donovan, MD, PhD
“Refugee Healthcare Quality Improvement Project”
Investigator: Steve Klotz, MD
 “Testing for Chagas in Victims of Kissing Bug Bites”
“Testing for Chagas in Victims of Kissing Bug Bites”
Investigator: Steve Klotz, MD
“Massive Bee Envenomation”
Investigator: Steve Klotz, MD
Current Clinical Trials
Faculty at the College of Medicine – Tucson also engage in clinical trials to develop new, more effective therapeutic remedies for infectious diseases. This often involves a research study conducted to evaluate a medical procedure or medical product, such as a drug. Not all UArizona studies involve drugs or interventions. Some studies use surveys or evaluate medical records to find new and better ways to help people. Others recruit healthy subjects, or controls, to better evaluate and compare results with those of non-healthy subjects.
Other clinical trials at the UArizona Health Sciences
How to Participate
Whether you are a potential clinical trial candidate as a patient or subject in the study, you would like to participate as a “healthy volunteer” for the control group, or you’re a research or clinical professional interested in collaborating with the research team — just contact the study coordinator or principal investigator listed for each individual clinical research study above.
You may also reach out directly to the office of the Division of Infectious Diseases, (520) 626-6887 — stephanie.abalos@bannerhealth.com — for additional information.


