As a faculty physician within the Division of Infectious Diseases, my research includes the following studies:
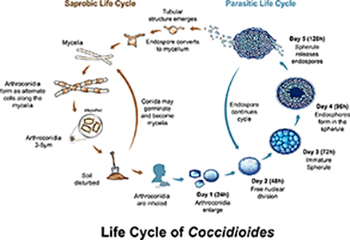 1. Antigen Detection for Diagnosis of Pulmonary and Disseminated Coccidioidomycosis Among Those with Compromised Immune Systems: The role of Coccidioidomycosis (or Valley Fever) antigen testing from serum and Bronchoalveolar lavage (BAL) samples is being evaluated. In addition, cross reactivity with other fungal tests including (1-3)-Beta-D-Glucan assay is being evaluated.
1. Antigen Detection for Diagnosis of Pulmonary and Disseminated Coccidioidomycosis Among Those with Compromised Immune Systems: The role of Coccidioidomycosis (or Valley Fever) antigen testing from serum and Bronchoalveolar lavage (BAL) samples is being evaluated. In addition, cross reactivity with other fungal tests including (1-3)-Beta-D-Glucan assay is being evaluated.
2. Prevention of Coccidioidomycosis Infection Among Liver Transplant Recipients - A Single Center Experience: The study has been completed and was recently presented at the World Transplant Congress. It evaluated the dose, duration, and efficacy of low dose versus standard dose fluconazole utilized for the prevention of post-transplant Coccidioidomycosis. The manuscript is being finalized.
3. Factors Correlated with Success in Graduate Medical Education: The purpose of this research is to identify the educational data that is correlated with performance in the Departments of Pediatrics and Medicine GME program in order to improve the residency application process.
4. Predictors of complicated urinary tract infections among renal transplant recipients: This is a retrospective review of the factors leading to the development of multi-drug resistance bacterial infections post renal transplantation.
5. Cardiac Device-Related Infections in Destination and Bridge to Heart Transplantation - A Single Center Experience: The goal of this study is to evaluate the risk factors, type of bacteria, perioperative antibiotic use, and other factors leading to device related infections in patients awaiting cardiac transplantation and those with advanced heart disease.
6. A Randomized, Open-Label Study to Evaluate the Safety and Efficacy of ABT-450/Ritonavir/ABT-267 (ABT-450/r/ABT-267) and ABT-333 Coadministered with Ribavirin (RBV) in Adults with Genotype 1 Chronic Hepatitis C Virus (HCV) Infection and Human Immunodeficiency Virus, Type 1 (HIV-1) Coinfection (TURQUOISE-I): The primary objectives of this study are to assess safety and to compare the SVR12 rates of co-formulated ABT-450, ritonavir and ABT-267 (ABT-450/r/ABT-267) and ABT-333 co-administered with ribavirin (RBV) for 12 or 24 weeks to the historical SVR rate of pegIFN and RBV in genotype (GT) 1 HCV-infected adults with HIV-1 coinfection.

